|
FUNGAL
SKIN INFECTIONS
|
|
|
|
Candidiasis is a superficial fungal infection, which is very common in infants and young children caused by the yeast-like fungus Candida albicans. C. Albicans is a commensal of the oral cavity, gastrointestinal tract and vagina. Predisposing factors It has long been recognized that the very old, the very young and the very ill are susceptible to oral thrush. The organism changes its behavior and becomes pathogenic under certain conditions
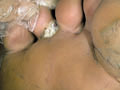
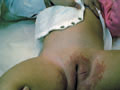
The clinical picture varies according to the site involved. These may be cutaneous, muco cutaneous or mucosal lesions. Skin Manifestations The intertriginous areas are common sites for Candida infection particularly in infants and young children due to occlusion and maceration of skin. The lesion appears as an erythematous eruption with smaller lesions on the periphery known as satellites. In adults and school age children the interdigital involvement is common causing inflammation and erosion. The nails may be infected leading to localized inflamed swollen peri-ungual tissue, where beads of pus can be squeezed from the lesion. The lesion may involve the tongue and most of the oral mucosa, giving the appearance of white mottled sheet. This may interfere with feeding of infants where they can‘t tolerate bottle-feeding or some acid food or beverages such as orange juice. In chronic cases the infection may extend to the angles of the mouth to the muco-cutaneous junction leading to fissuring and even bleeding from the angle of the mouth. Candidal Paronychia This condition is more common in adults especially housemaids whose hands are continuously immersed in water. The lesion begins as painful inflammation of the side of the nail. Beads of pus may be expressed from the lesion. Secondary infection by staphylococci is common. Vaginal candidiasis This is common also during pregnancy and in infancy. Vaginal mucosa becomes red macerated and may be covered with white membrane. This may be accompanied by vaginal whitish creamy discharge .The adjacent skin may be involved and becomes red, scaly and may show some satellite pustules.
Normal subjects have antibodies, humoral and cellular, to Candida albicans and to other Candida species. The term "candida allergy" is also used to describe a variety of symptoms ranging from headache to malaise and depression secondary to colonization of the gastrointestinal tract with the yeast. Manifestations:
Treatment of candidiasis
Treatment of Oral Candidiasis In infants suspensions of Nystatin, Amphotericin or Miconazole gel applied several times a day are usually adequate for treating oral thrush. In the adult patient, removal of the dentures at night and careful hygiene is important. Frequent sucking of Amphotericin lozenges, which lack the bitter taste of Nystatin. Amphotericin tablets are also effective. Daktarin gel and oral suspensions of Nystatin. The duration of the treatment varies according to the type and extent of the skin lesion. Treatment duration is about 10-14 days. This may be enough in acute cases but in chronic hyperplastic candidiasis it must be continued for many months. Angular Candidiasis - imidazole cream or ointment may be enough . Pufexamac(Flogocid) is cheaper and gives good results. Oral ketoconazoles, itraconazoles usually give good results especially when Candida infection is widespread. Congenital candidiasis - localized lesions of candidiasis can be treated by topical preparations. Systemic Candidiasis - Amphotericin, Flucytosine , Fluconazole, orally and paranterally may be required to control systemic infections .
Candidiasis may appear on newborn few weeks after labor in two forms. Skin and mucous membrane lesions : may result from infection from infected genital tract of the mother during labor . Oral candidiasis may appear alone during suckling .
Chorio-amnionitis may follow infection of the mother‘s genital tract. Candida infection affects skin and internal organs such as the lungs and gastrointestinal tract. This type is serious and there may be high mortality rate. Clinical Features Skin manifestations The face and chest are the first affected by the rash, which generally spreads over the next few days after delivery. The primary skin lesions are diffuse, pinkish, maculopapular eruption which is present at birth or appear later after few hours. The lesions are typically discrete vesicles or pustules on an erythematous base. The lesions generally progress to a vesicular phase, and then either to a pustular or a bullous phase, over a period of 1 to 3 days. More or less the whole skin surface may be affected, including the palms and soles. Oral involvement is usually absent, and the napkin area tends to be spared, at least initially. When infection is localized to the skin, the rash clears within a week with an appropriate topical therapy, with post inflammatory desquamation. The general condition of the child is usually not affected.
Candida may invade internal organs mainly the gastrointestinal tract and the respiratory system leading to premature babies and high mortality rates . Such widespread skin infections are believed to follow contamination of the skin surface during birth and to the high incidence of intra-uterine infection or vaginal candidiasis.
FUNGAL
INFECTIONS Different mould saprophytes may colonize either normal skin or devitalized skin tissue causing fungal infection. Saprophytes normally colonize the scalp and toe-clefts.
Treatment Correction of local precipitating factors such as maceration, occlusive dressings. Topical antifungal agents may be required. It may be necessary to use intravenous Amphotericin B in some systemic cases. The newer Azole agents such as Ketoconazole, Fluconazole or Itraconazole may be helpful and are more convenient.
OTOMYCOSIS This is a chronic fungal infection of the external auditory canal . Fungi mainly Asperigillus species may be isolated from swabs or scrapings where these fungi may be saprophytic or pathogenic . Clinical Picture The external ear becomes inflamed, painful, itchy and weeping with serosanguinous discharge . In advanced cases of true mycotic otitis, an overgrowth of fungal hyphae may produce a mass of white material which appears as a damp cotton wool lodged in the external canal. Asperigillus Niger is the causative organism where the mat of fungus is often covered by black fruiting heads. In severe cases necrotic otitis externa may develop . This form may spread to involve other sites including the middle ear and the mastoids. The pinna may be the site of several mycotic diseases including Chromomycosis , Sporotrichosis and Tinea but such infections usually spare the external auditory meatus. Diagnosis of mycotic otitis externa Smear: swab taken gently from the ear. Culture to detect the type of mould. Treatment
Sporotrichosis is a chronic fungal infectious disease caused by Sporothrix Schenkeii. The organism lives as a saprophyte in grasses and plants where an accidental injury will facilitate the inoculation of the organism into the skin . The disease is more common in hot and humid environments . Clinical Manifestation
Treatment Treatment depends on different factors. The advice of an experienced practitioner in deep mycoses may be required in some cases. Potassium iodide in large doses by mouth is effective in the localized types and should be continued for 3-4 weeks after clinical cure. It is the drug of choice for the cutaneous form. The adult dose may be 40 drops of potassium iodide daily. The treatment course is usually from 6-8 weeks. A recommended schedule is 5 drops initially, then increasing to 50 drops of saturated KI three times a day. Patient tolerance may require a lower maximum dose . Itraconazole in doses of 100-200 mg daily is effective but it appears that the length of treatment is not significantly different to that used with potassium iodide. It may, however, be useful in patients who do not respond to the latter or in systemic cases. Intravenous Amphotericin B or Miconazole may also be helpful. Ketoconazole produces variable results in Sporotrichosis and in many cases there is no response to this drug.
This is a systemic deep fungal infection caused by Coccidioides immitis. Modes of Infection The organism is present in the soil, vegetables, and especially fruits . Infection is by inhalation of dust contaminated with the spores of the fungus. The disease is wide spread in endemic areas especially in dry windy summer months. Clinical Manifestations The incubation period may be from few days to several weeks. General manifestations Mild respiratory symptoms with non-specific symptoms such as high fever, chills, night sweating , headache , backache, malaise and bronchopneumonia. Skin manifestations Generalized maculopapular eruption simulating drug eruption or measles appears in infants and children which manifests early with the onset of infection. Erythema nodosum on the chins, thigh, buttocks, may present after the respiratory symptoms subside . Disseminated type The disease is usually self-limiting, where most cases recover spontaneously. Few cases pass to the disseminated form from the localized lesions to lungs, bones, viscera, and meninges. Skin lesion presents with subcutaneous abscess and forming draining sinuses. Healing is by tissue destruction and scarring . Diagnosis: Skin biopsy is diagnostic Treatment Intravenous Amphotericin , 0.25 mg/kgm body weight . The dose should be increased gradually where 0.1mg/kgm may be the optimum daily dose .
This is a systemic deep fungal infection . Types of Blastomycosis
This is a deep fungal infection caused by Blastomyces dermatidis, which is endemic in North America . Clinical manifestations
Differential Diagnosis
Treatment Amphotericin B intravenously is an effective medication.
Clinical manifestations
Treatment Amphotericin B is an effective medication .
Chromoblastomycosis is a deep fungal infection caused by various fungi mainly Cladosporium carrionii and Philaphora verrucosa . The lesions affect usually the feet or lower extremities in patients walking barefooted . Clinical manifestations
Histopathology The histopathological picture includes Granulomatous reaction. Pseudotubercles containing giant cells and focal cell infiltrate . The fungus appears as brown, spherical clusters with thick dark cell wall and coarsely pigmented granular cytoplasm . Treatment Surgical excision and grafting. Intravenous Amphotericin B may have some effect . Intralesional Amphotericin B.
Histoplasmosis is a systemic mycoses caused by the saprophyte, histoplasmosis capsulatum, which is present in the soil. Dissemination of infection to the skin is infrequent. The disease has a serious prognosis in children . Clinical Types
Skin lesions may appear in crops leading to ulcers or umbulicated nodules and papules. Secondary bacterial infection is common , where pyoderma, furuncles and abscesses may involve the infected areas. Treatment Amphotericin B is the drug of choice. Sulphonamides.
Cryptoccosis is a systemic mycoses caused by Creptococcus neoformans which is present in the soil, dust and as a saprophyte on the human skin. Clinical manifestations Primary pulmonary type - manifests with mild cough ,chest pain and fever. The disease can be diagnosed radiologically at this stage. Central nervous system - manifestations are due to dissemination of the disease causing intracranial hypertension. These include restlessness, depression, hallucination, headache, vertigo, nausea and vomiting. Skin manifestation - dissemination of the disease to the skin presents with indolent rubbery acniform papules or pustules on the face. Ulceration and granulomatous lesions may occur. Treatment Amphotericin B usually gives good results.
Nocardiosis is a systemic mycoses caused by Nocardia asteroids and N. Brazilians. Clinical manifestations Pulmonary type - presents with cough, anorexia, night sweats and weight loss. Skin manifestations - these are variable which may be multiple abscesses draining from the chest lesions, vesicular eruption or with cutaneous nodule at the site of inoculation of the causative fungi. Treatment Sulphonamides Other types of antibiotics such as penicillin and Tetracyclines may be effective.
MYCETOMA Mycetoma is a systemic mycoses caused by the group Streptomyces Somaliens, S. Madura and other species such as Nocardia group. The disease appears mainly in the western Hemisphere, South America and Africa. Clinical manifestations Skin lesions present with subcutaneous swelling on the interdigital spaces, buttocks, and chest or on other areas. The nodules are painless, and indolent. Ulceration may follow with draining sinuses of the foot. Treatment Treatment depends on the type of the lesion and the causative organism. In the early stage removal of the affected area or even amputation of the affected limb in severe and extensive lesions. Sulfadiazine in Nocardia lesions may be effective. Sulfisoxazole(Gantrisine) and Sulfones.
Rhinosporidosis is a polypoid disease that involves mainly the nasal mucosa. Young children and adults are commonly affected. The disease is caused by Rhinospedium seeberi. The disease is endemic in India, Ceylon, South America, Italy and other parts of the world . Clinical manifestations The lesions affect mainly the nasal mucosa. Other areas involved are the lacrimal sac, ears, vulva and penis. Pinkish, papillomatous, fissured lesions develop which become fissured and bleed later one. Rectal and vaginal lesions present with the same manifestations. Treatment Electrodessication.
|
| Contents | << Previous Chapter | Next Chapter >> | Search |